FDA BRIEF: Week of October 30, 2017
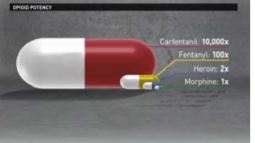
Statement from FDA Commissioner Scott Gottlieb, M.D., on new strategies for addressing the crisis of opioid addiction through innovation in packaging, storage and disposal
Steps to reduce the scope and human tragedy of opioid addiction epidemic
- New ways to work more creatively with public and private partners
- Reduce exposure to opioid drugs
- Proposer prescriptions and duration of treatment
Packaging innovations
- Packaged in a manner that limits the number of pills dispensed
- Easier to track the number of doses that have been taken
- Improve storage and encourage prompt disposal
- Allow health care providers, pharmacists or family members to monitor patient use of prescription opioids
public workshop, Dec. 11-12, 2017, to advance this issue
 FDA’s Role in Medical Device Cybersecurity
FDA’s Role in Medical Device Cybersecurity
Breach potentially impacting safety and effectiveness of a medical device can threaten patient health
- CDRH coordinated approach of vigilance, responsiveness, resilience, and recovery
- Total product lifecycle approach- starting at the product design phase to planning how to reduce the likelihood of future risks
- Encouraging medical device manufacturers to proactively update and patch devices in a safe and timely manner
- Need to balance protecting patient safety and promoting the development of innovative technologies and improved device performance
- Published guidances for manufacturers
- Planning to address cybersecurity risks is as essential to the device development process as coming up with a novel new product
prevalent myths concerning FDA
 FDA takes unprecedented step toward more efficient global pharmaceutical manufacturing inspections
FDA takes unprecedented step toward more efficient global pharmaceutical manufacturing inspections
FDA will recognize eight EU drug regulatory authorities as capable of conducting inspections of manufacturing facilities that meet FDA requirements
- Austria, Croatia, France, Italy, Malta, Spain, Sweden, UK
- Implementation of US-EU Mutual Recognition Agreement
- Will now rely on the inspectional data obtained by these eight regulatory agencies
EU Commission determined (6/17) that the FDA “has the capability, capacity and procedures in place to carry out GMP inspections at a level equivalent to the EU.”
- Enables the FDA and the EU to avoid duplication of drug inspections
- Allows regulators to devote resources to other high risk manufacturing facilities
FDA warns companies marketing unproven products, derived from marijuana, that claim to treat or cure cancer
Increasing concern with cannabidiol (CBD) products claiming to treat or cure cancer
- Marketed in a variety of product types e.g. oil drops, capsules, syrups, teas
- Not approved for any indication
- Substances containing marijuana components treated like any other products making unproven claims to shrink cancer tumor
Warning letters to four companies for unsubstantiated claims
- Greenroads Health
- Natural Alchemist
- That’s Natural! Marketing and Consulting
- Stanley Brothers Social Enterprises LLC
 Health Literacy: Getting the Info You Need
Health Literacy: Getting the Info You Need
Health literacy is the ability to get and understand information on health issues and medical services so that patients you can make informed decisions
- ~ 12% US adults have skills to manage health and prevent disease
- Difficult to improve their health
FDA Promotes Health Literacy
- Plain language for clear communications
- Patient package inserts, instructions for use, and Medication Guides
- Drug Safety Communications
- Recalls and Withdrawals
- Free online resources
- FDA Patient Network
- FDA Consumer Updates
Image credits: FDA, EMA
