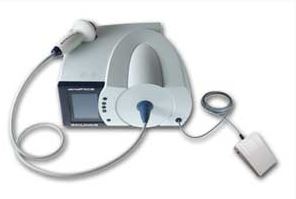
DERMAPACE System
Sanuwave, Inc.
INDICATION FOR USE: To provide acoustic pressure shockwaves in the treatment of chronic, full-thickness diabetic foot ulcers with wound areas measuring no larger than 16 cm2 , which extend through the epidermis, dermis, tendon, or capsule, but without bone exposure.
The dermaPACE System is indicated for adult (22 years and older), diabetic patients presenting with diabetic foot ulcers greater than 30 days in duration and is indicated for use in conjunction with standard diabetic ulcer care.
ADDRESSING UNMET NEED:
- About 25 percent of people with diabetes will experience a foot ulcer in their lifetime; leading cause of lower limb amputations
- Additional option for successfully treating and healing ulcer wounds may help prevent lower limb amputations
GENERIC TYPE OF DEVICE:
- Extracorporeal shockwave device for treatment of chronic wounds is a prescription device that focuses acoustic shock waves onto the dermal tissue. The shock waves are generated inside the device and transferred to thevbody using an acoustic interface.
EFFECTIVENESS AND SAFETY:
- Two multi-center, randomized, double-blind studies, n= 336 diabetic patients receiving either usual care plus Dermapace System shock wave therapy vs. usual care plus non-working (sham) shock wave therapy
- Both patient groups included those with poorly controlled and well-controlled blood glucose levels
- Increase in wound healing at 24 weeks, 44% vs. 30% wound closure rate
- Most common side effects: Pain, local bruising and numbness, migraines, nausea, fainting, wound infection, infection beyond the wound (cellulitis, osteomyelitis) and fever
REGULATORY PATHWAY: De Novo
- Initial PMA submission not approved
- Resubmission as De Novo
- Regulation Number: 21 CFR 878.4685
- Regulation Name: Extracorporeal shock wave device for treatment of chronic wounds
- Regulatory Class: Class II
- Product Code: PZL
RISKS AND MITIGATIONS:
- Adverse tissue reaction – Biocompatibility evaluation
- Infection – Reprocessing validation, Labeling
- Inadequate healing – Labeling
- Device failure / malfunction – Non-clinical performance testing, Electrical safety testing, Electromagnetic compatibility (EMC) testing, Use life testing, Software verification, validation, and hazard analysis, Labeling
- Hearing loss – Non-clinical performance testing, Labeling
REIMBURSEMENT: Pending
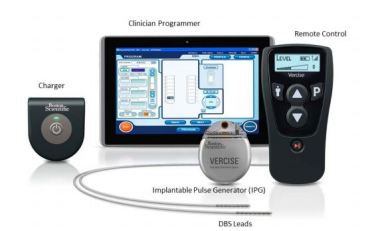 VERCISE Deep Brain Stimulation (DBS) System
VERCISE Deep Brain Stimulation (DBS) System
Boston Scientific Corporation
INDICATION FOR USE: In bilateral stimulation of the subthalamic nucleus (STN) as an adjunctive therapy in reducing some of the symptoms of moderate to advanced levodopa-responsive Parkinson’s disease (PD) that are not adequately controlled with medication.
ADDRESSING UNMET NEED:
- At present there is no cure for PD
- Additional surgical treatment option focused on management of symptoms that best meets expectations and lifestyle
- Adjunct to therapy towards reducing the motor complications of subjects with PD
DEVICE DESCRIPTION:
- Includes a Stimulator with DBS Leads for stimulation of selected targets (i.e., the subthalamic nucleus) in the brain.
- DBS Extensions are used to connect the DBS Leads to the Stimulator implanted near the clavicle.
- The Vercise DBS System utilizes current steering across eight contacts per DBS Lead,
which is intended to provide precise positioning of stimulation. - The Stimulator is controlled by a handheld Remote Control, and can be programmed by a Clinician Programmer using the Bionic Navigator™ Software.
- Periodically, the rechargeable Stimulator battery must be replenished with a radiofrequency (RF) charging device provided in the Charging Kit.
EFFECTIVENESS:
- Study on bilateral DBS of the STN, n=160 pre-specified interim analysis; active vs control, 12 weeks
- Primary endpoint: Mean change from baseline in mean number of waking hours per day with good symptom control and no troublesome dyskinesia as measured on the PD diary, with no increase in antiparkinsonian medications – mean difference of 3.03 ± 4.2 hours, p < 0.001
- Secondary endpoints: UPDRS III scores in the stim on/meds off condition, PDQ-39, Modified Schwab and England, Global Impression of Change as assessed by clinician and Treatment Satisfaction
- UPDRS III score: Mean 30% improvement (12.02 ± 11.42 points) was noted in UDPRS III scores (stim on/meds off)
- Statistically significant improvement in quality of life: Based on PDQ-39 and modified Schwab and England scales
SAFETY:
- Infection: Most commonly reported serious adverse event associated with device-hardware/procedure
REGULATORY PATHWAY: PMA
- Device Generic Name: Stimulator, Electrical, Implanted, for Parkinsonian Tremor
- Device Procode: NHL
REIMBURSEMENT:
- DBS Devices assigned ICD-10 daignosis and procedure codes
- HCPCS II Device Codes
- Device C-Codes and Device Edits
- CPT Procedure Codes
- MS-DRG Assignments

HEMOBLAST Bellows
Biom’Up SA
INDICATION FOR USE: In surgical procedures as an adjunct to hemostasis when control of minimal, mild, and moderate bleeding by conventional procedures is ineffective or impractical, except in neurosurgical, ophthalmic and urological procedures.
DEVICE DESCRIPTION:
- Consists of hemostatic powder (HEMOBLAST™ Bellows Hemostatic Powder) supplied in an applicator system incorporating a bellows design
- Powder is dry, sterilized, biocompatible, and non-pyrogenic. No intraoperative preparation, mixing, or heating is required. absorbs in vivo over a 4-week period
- Powder composed predominantly of highly purified porcine collagen (with glucose) with smaller amounts of bovine chondroitin sulfate (CS) and human pooled plasma derived thrombin.
- Bellows Applicator contains the Powder and is sterilized using gamma-sterilization
- Nozzle Extension serves to assist in the delivery of the Powder, during surgery
EFFECTIVENESS AND SAFETY:
- Prospective, multicenter, single-arm pilot clinical investigation, to evaluate Surface Bleeding Severity Scale (“SBSS”), n=31 undergoing orthopedic and abdominal surgeries with associated bleeding sites
- Statistical superiority in achieving hemostasis at 3 and 6 minutes vs. standard of care
- Safety profile similar to standard of care
REGULATORY PATHWAY: Combination product, PMA
- Regulation number: 878.4490
- Product Code: PMX
- Generic name: Absorbable Collagen Hemostatic Agent With Thrombin
REIMBURSEMENT:
- Pending
- Precedents for reimbursement for absorbable collagen hemostatic
Image credits: Sanuwave, Boston Scientific, Biom’Up