News & Views: 2019 Shutdown and Funding Lapse, Breakthrough Devices, Safer Technologies Program, Software Precertification Program Model v1.0, Sentinel System Roadmap
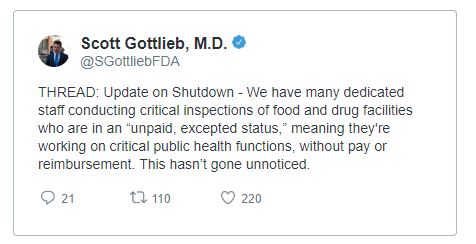
FDA 2019 Lapse in Funding Information
Agency operations continue to the extent permitted by law necessary to address imminent threats
- Handle and respond to emergencies
- Mission critical surveillance for significant safety concerns with medical devices and other medical products
Activities funded by carryover user fee funds
- NO acceptance of regulatory submissions for FY 2019 that require a fee payment and that are submitted during the lapse period
READ ………………………………………………………………………………
Breakthrough Devices Program
 Goal
Goal
- Provide timely access to patients and health care providers by speeding up development, assessment, and review (PMA, 510(k), De Novo)
- Replaces Expedited Access Pathway and Priority Review for medical devices
Benefits
- Efficient FDA interaction during premarket review phase
- Prioritized review of submission
READ ………………………………………………………………………………

Safer Technologies Program (STeP)
As part of Medical Device Safety Action Plan, STeP is for devices that treat less serious conditions and innovate on safety
- For devices that do not otherwise meet ‘breakthrough device’ criteria
- Has the potential to be significantly safer than currently available alternative treatments or diagnostics
- Additional details to be provided later
Substantial safety innovations could include
- Reduce occurrence of serious adverse event or other safety issue
- Address known device failure mode or common user error
- Provide for significant safety advantages for users
READ ………………………………………………………………………………
 Digital Health Software Precertification (Pre-Cert) Program: v1.0 Working Model
Digital Health Software Precertification (Pre-Cert) Program: v1.0 Working Model
Objective
- Streamlined and efficient regulatory oversight of software-based medical devices
- Manufacturers who have demonstrated robust culture of quality and organizational excellence
- Manufacturers who are committed to monitoring real-world post-marketing performance
Proposed key components
- Excellence Appraisal: Five excellence principles of patient safety, product quality, clinical responsibility, cybersecurity responsibility, and proactive culture
- Review: De Novo classification process with review corresponding to excellence principles
- Streamlined Review: Interactive review of software product with special controls and TPLC approach
- Real-world Performance: Type of information about how software product is performing with patients and emerging risks
READ ………………………………………………………………………………

Sentinel System – Five-Year Strategy 2019-2023
Sentinel System
- Core feature of medical product post-market safety surveillance armamentarium
- Proactively monitors product safety, methodological innovation, real-world evidence
- Roadmap to focus on innovations emerging from new data science disciplines (NLP, ML), , expand access and use of EHRs
Five Strategic Aims
- Enhance and expand foundation, including data, infrastructure, operations and technology
- Augment safety analysis capabilities using advanced data science, signal detection
- Accelerate access to and broaden use of real-world data for real-world evidence
- Broaden stakeholder ecosystem
- Disseminate knowledge and advance regulatory science
READ ………………………………………………………………………………
Image credits: FDA